Oxygen is a life-supporting gas. Nearly all living things need oxygen to stay alive. Oxygen combines with other chemicals in plant and animal cells to produce energy needed for life processes. Oxygen is also needed to make most fuels burn. During the burning process, oxygen combines with the fuel in a chemical reaction. Heat is released during the burning process.

Oxygen is also a chemical element. It is one of the most plentiful chemical elements on Earth. It makes up about a fifth of the volume of air. Nitrogen makes up most of the other four-fifths. Oxygen is also found in Earth’s crust and in water. This oxygen is not pure but is combined with other elements. On the average, 100 pounds of Earth’s crust contains about 47 pounds of oxygen. Nearly half of the weight of most rocks and minerals is oxygen. Every 100 pounds of water contains about 89 pounds of oxygen. Hydrogen makes up the other 11 pounds of water.
Chemical properties.
Oxygen has an atomic number (number of protons in its nucleus) of 8. Its relative atomic mass is 15.9994. An element’s relative atomic mass equals its mass (amount of matter) divided by 1/12 of the mass of carbon 12, the most abundant form of carbon. The chemical symbol of oxygen is O. Chemists classify oxygen as a nonmetal. For information on the position of oxygen on the periodic table, see the article Periodic table.
Ordinary oxygen molecules, such as those that make up about 20 percent of the atmosphere, have two oxygen atoms bonded together. This ordinary oxygen is colorless, odorless, and tasteless. Oxygen molecules that have three oxygen atoms make up a gas called ozone. Oxygen combines with many other elements, forming compounds called oxides (see Oxide).
Ordinary oxygen changes to a pale blue liquid when cooled to its boiling point. Oxygen liquefies (becomes a liquid) at –182.962 °C at atmospheric pressure. Oxygen liquefies at a higher temperature when the pressure is increased. At a pressure of 731.4 pounds per square inch (51 kilograms per square centimeter, or 5.043 megapascals), oxygen liquefies at –118.55 °C. These values of temperature and pressure are oxygen’s critical temperature and pressure. It is impossible to liquefy oxygen at a higher temperature at any pressure. Liquid oxygen is magnetic. It can be held between the poles of a strong magnet. It freezes at –218.4 °C.
How oxygen supports life.
Only a few kinds of living things, including certain germs, can live without free (chemically uncombined) oxygen. Human beings and other land animals get oxygen from the air. Fish and most other water animals get dissolved oxygen from water.
Free oxygen enters a person’s bloodstream through the lungs. It enters a fish’s bloodstream through the gills. The blood carries oxygen to the cells of the body. In the cells, oxygen combines with chemicals obtained from food. Energy produced during this process makes it possible for each cell to perform its function in the body. Carbon dioxide is produced in the cells as a waste product (see Respiration).
Plant cells use oxygen in much the same way that animal cells do. Plant cells also make oxygen in the process of photosynthesis (see Photosynthesis). During this process, the cells use the energy of sunlight to make sugar from carbon dioxide and water. Oxygen is produced as a by-product and is released into the atmosphere. Single-celled organisms called cyanobacteria also produce oxygen via photosynthesis. Cyanobacteria appeared many millions of years before multicellular plants. Scientists believe that all of the air’s oxygen has been formed by photosynthesis over billions of years. Geologists studying ancient soil layers have observed a large increase in iron oxide compounds (rust) dating from about 2.4 billion years ago. These layers of oxygen compounds indicate the time in Earth’s history when free oxygen from photosynthesis began to build up in the atmosphere. Scientists believe that this time, also known as the Great Oxidation Event, helped allow more complex organisms to evolve.
Other uses of oxygen.
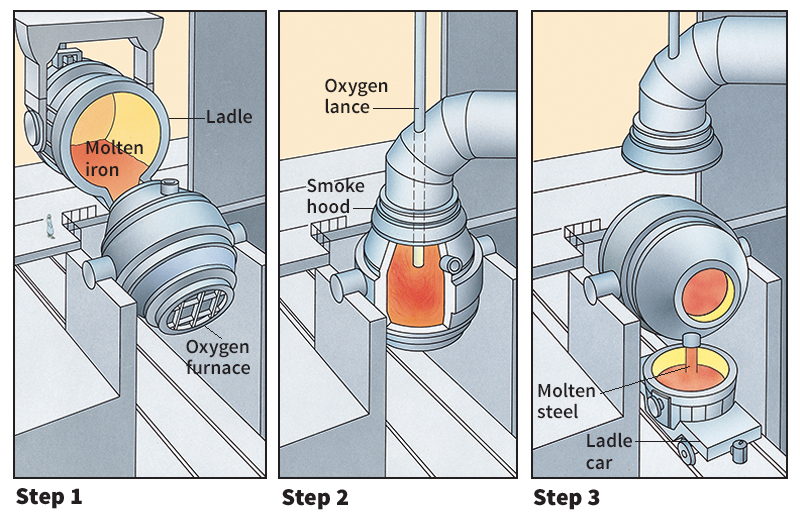
Oxygen has many uses in industry. Some steel is manufactured by the basic oxygen process. In this process, a stream of high-pressure oxygen blasts down on melted pig iron. The oxygen burns out impurities. Welders mix oxygen with fuel in their torches to produce a hot flame. The flames burns at a temperature of about 6000 °F (3300 °C).
Liquid oxygen, called LOX, is used in rockets propelled by liquid fuels. LOX combines with various fuels, including kerosene and liquid hydrogen, to produce the rocket’s thrust (pushing force). LOX is also mixed with other fuels to make explosives for blasting.
Making oxygen.
Most commercial oxygen is distilled from liquid air. During the distillation process, the nitrogen boils before the oxygen does, because nitrogen has a lower boiling point. As the nitrogen boils away, the liquid air is left with a greater concentration of oxygen. Commercial oxygen is stored in steel tanks at a pressure of about 2,000 pounds per square inch (140 kilograms per square centimeter, or 14 megapascals). That pressure is more than a hundred times the pressure of the atmosphere.
Small amounts of oxygen can be made by heating potassium chlorate. A little manganese dioxide added to the potassium chlorate speeds up the formation of oxygen.
History.
Oxygen was discovered by two chemists working independently. They were Carl Scheele of Sweden and Joseph Priestley of England. Scheele’s laboratory notes show that he prepared oxygen in the early 1770’s by heating various compounds, including saltpeter and mercuric oxide. Scheele’s experiments were not published until 1777. Priestley, however, had his experiments with oxygen published in 1775. Further observations concerning the gas were published in 1777. Priestley described how he prepared oxygen in 1774 by heating mercuric oxide.
Scheele called oxygen fire air. Priestley called it dephlogisticated air. In 1779, the French chemist Antoine Lavoisier named the gas oxygen. The word means acid producer. Lavoisier and others had found that oxygen is a part of several acids. Lavoisier incorrectly reasoned that oxygen is needed to make all acids. He combined the Greek words oxys (meaning sharp or acid) and gignomai (meaning produce) to form the French word oxygene. This word is oxygen in English.

